In 2026, Isotonix remains under intense regulatory and legal scrutiny, a situation that has created significant consumer confusion and concern. The phrase “Isotonix lawsuit” frequently surfaces in online searches, often blending separate legal actions, a major FDA warning letter, and ongoing discussions about the brand’s multi-level marketing (MLM) structure. This comprehensive guide clarifies the current landscape, separating verified facts from widespread misinformation.
Isotonix is a brand of powdered dietary supplements, owned and distributed by Market America. The brand markets its products using an “isotonic delivery system,” claiming that when mixed with water, the solution matches the body’s own fluids, allowing for faster and more efficient nutrient absorption. These supplements are sold exclusively through Market America’s network of independent distributors, a model that is itself a central part of the controversy.
The legal and regulatory challenges are twofold. First, there is significant regulatory action from the U.S. Food and Drug Administration (FDA), documented in an official 2020 warning letter. Second, there are historical civil litigation allegations, primarily a 2017 class-action lawsuit that accused Market America of operating an illegal pyramid scheme. It is crucial to understand that, as of early 2026, there is no major active, certified class-action lawsuit specifically targeting Isotonix supplements for consumer injury or false advertising that is proceeding publicly in court.
This article, reviewed for accuracy against FDA documents and legal records, provides a clear, evidence-based breakdown of what has happened, what the current status is, and what rights consumers and distributors have. We will dissect the FDA’s findings, explain the outcome of past litigation, and offer practical steps for those affected.
What Is Isotonix? Brand Background & Business Model
The Science Behind Isotonix Supplements
Isotonix distinguishes itself from traditional pill or capsule vitamins by its delivery format. The products are sold as powders that consumers mix with water to create an “isotonic” solution—one with a similar salt and sugar concentration as blood plasma. The core marketing claim is that this allows nutrients to pass through the stomach and be absorbed in the small intestine more quickly and completely, a process the company calls “isotonic-capable” delivery.
The product line includes popular items like Isotonix OPC-3 (an antioxidant blend), Isotonix Multivitamin, Activated B-Complex, and Heart Health Essential Omega III. However, critics and regulators have questioned the scientific substantiation for the superiority of this delivery method over other forms, noting that bold absorption claims may not be fully backed by independent, peer-reviewed research.
Market America’s Multi-Level Marketing Structure
Market America operates on a multi-level marketing (MLM) model, also known as network marketing. In this structure, independent distributors (called “UnFranchise Owners”) earn income through commissions on personal product sales and, significantly, on sales made by other distributors they recruit into their “downline”.
The legal controversy often centers on whether the primary focus is on genuine retail sales to consumers or on recruiting new participants. Legitimate MLMs are legal, but the FTC states that a venture becomes an illegal pyramid scheme if it “promises participants payment primarily for recruiting others rather than for the sale of goods or services”. Allegations that Market America’s model emphasizes recruitment—where the real money is made from selling starter kits and inventory to new recruits rather than to end consumers form the basis of past lawsuits.
The Isotonix Lawsuit Explained: 2026 Legal Landscape
Is There an Active Class Action Lawsuit Against Isotonix in 2026?
As of January 2026, there is no major, active class-action lawsuit against Isotonix or Market America that is publicly progressing in court regarding product efficacy, safety, or false advertising. Public court records do not show new, certified class actions filed on behalf of consumers.
Much of the online search interest stems from confusion between three distinct issues:
- Regulatory Action: The FDA’s 2020 warning letter (discussed below).
- Past Civil Litigation: A 2017 pyramid scheme class action that was sent to private arbitration.
- Consumer Complaints: General grievances shared on forums and social media about product results or business practices.
FDA Warning Letter 2020: Specific Violations Documented
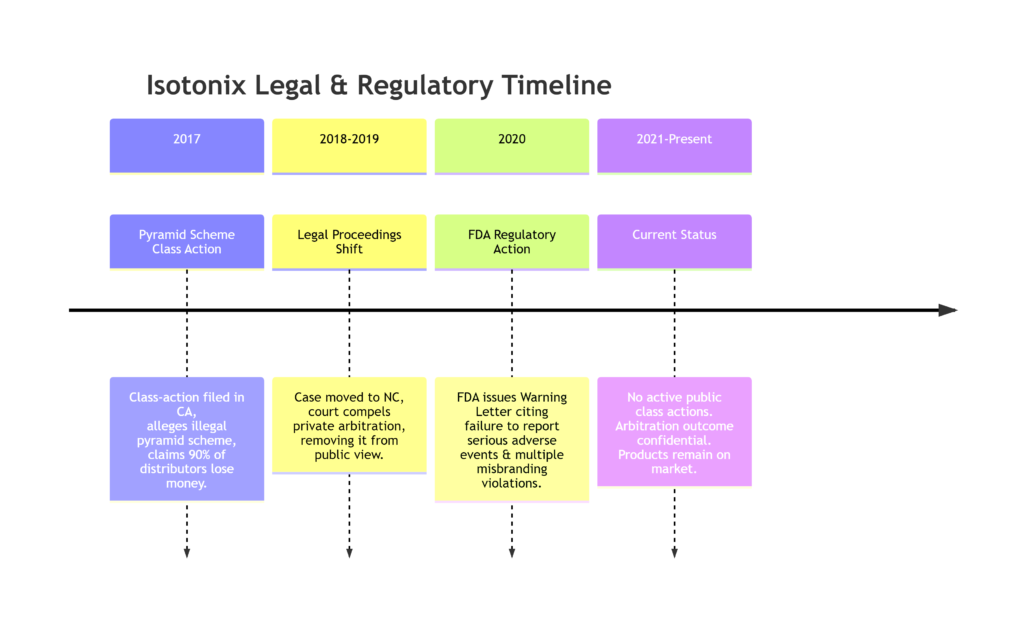
The most concrete regulatory action is a Warning Letter the FDA issued to Market America on February 12, 2020. This followed an inspection of the company’s facility. The letter cited serious violations of the Federal Food, Drug, and Cosmetic Act:
- Failure to Report Serious Adverse Events: The FDA stated Market America received but did not report two serious adverse event incidents requiring hospitalization. One involved a consumer who needed inpatient care for symptoms like abdominal pain, vomiting, dizziness, and weakness after using a “TLS 21-Day Challenge Kit,” which included Isotonix OPC-3. Federal law requires such reports within 15 business days.
- Misbranding of Multiple Isotonix Products: Several products were labeled incorrectly.
- Isotonix OPC-3: Listed an incorrect serving size (1 capful) when directions recommended 2 capfuls initially.
- Multivitamin & Multivitamin with Iron: Declared copper in “mcg” instead of “mg” and used a non-standard name for an herbal ingredient.
- Activated B-Complex & Heart Health Omega III: Included nutrients in amounts that could be declared as zero or used improper labeling formats.
- General Labeling: The FDA found “intervening material” within the official Supplement Facts box on several labels, which is not permitted.
Pyramid Scheme Allegations and MLM Litigation
In May 2017, a class-action lawsuit (Yang et al. v. Market America) was filed in California federal court. The plaintiffs, distributors, alleged Market America was an illegal pyramid scheme that violated federal and state racketeering (RICO) laws. The complaint claimed that over 90% of distributors lost money and that the company’s “two-year blueprint” for success was deceptive, as income was dependent on relentless recruitment rather than product sales.
Crucial Outcome: Market America invoked an arbitration clause in its distributor agreement. The case was moved to North Carolina and, by 2018-2019, a federal judge granted the company’s petition to compel arbitration. This means the dispute was moved out of the public court system and into private, confidential arbitration. As a result, there is no public verdict or settlement available, and the status of any individual claims is unknown.
State Attorney General Actions
While the 2017 class action was the most prominent litigation, the allegations put Market America on the radar of consumer watchdog groups. Organizations like TruthInAdvertising.org (TINA.org) have investigated and published reports on the company’s marketing practices, particularly regarding deceptive income claims made to potential distributors. Such scrutiny can sometimes precede or coincide with inquiries from state Attorneys General, though no specific, public state AG action against Market America over Isotonix is documented in the available sources.
Documented Allegations: Health Claims, Side Effects & Consumer Complaints
False Advertising and Unsubstantiated Health Claims
A persistent allegation is that Isotonix engages in misleading marketing. Critics argue that claims of “superior absorption” and “fast results” are not adequately supported by robust, independent clinical studies. The FDA warning letter focused on labeling violations rather than explicitly on these efficacy claims. However, under the FTC Act, companies must have competent and reliable scientific evidence to back any objective health or performance claims they make. The line between permissible “structure-function” claims (e.g., “supports immune health”) and impermissible disease treatment claims (e.g., “treats COVID-19 or diabetes”) is also a common area of regulatory risk for supplement brands.
Reported Side Effects and Adverse Events
The FDA’s warning letter provides the only officially documented serious adverse events linked to a product bundle containing Isotonix. Beyond this, side effect reports are largely anecdotal, found in consumer reviews and forums. Commonly mentioned issues include:
- Digestive discomfort (nausea, stomach pain)
- Allergic reactions (itching, rash)
- Headaches and dizziness
It is critical to note that adverse event reports do not prove causation. An event can be reported even if the supplement is not the definitive cause. However, the FDA’s finding that Market America failed to report serious events is a significant compliance failure that raises safety monitoring concerns.
Ingredient Transparency Issues
The FDA’s 2020 letter cited specific labeling inaccuracies, such as an incorrect botanical name (“lo han” vs. “luo han guo”) and failure to identify the plant part used for botanical ingredients in OPC-3. While not allegations of contamination, these are violations of the federal requirement that supplement labels be truthful and not misleading. Accurate labeling is fundamental to consumer trust and informed choice.
Timeline: Isotonix Legal and Regulatory History
The legal story of Isotonix is not a single event but a series of regulatory and legal challenges over the past decade.
Consumer Rights: What to Do If You’ve Used Isotonix Products
How to Report Adverse Effects
If you believe you have experienced a serious side effect from any supplement, you should report it directly to the FDA. This is a crucial right that also contributes to public safety monitoring.
- Process: Use the FDA’s MedWatch Voluntary Reporting Form online or by mail.
- Information to Provide: Include details of the product (name, lot number), the adverse effect, your contact information, and your healthcare provider’s information.
- Why Report: Your report helps the FDA identify potential patterns of harm that may not be visible to individual consumers or even the company.
Seeking Refunds or Compensation
- Company Policy: First, review Market America’s official return policy for the channel through which you purchased.
- Credit Card Dispute: If you purchased with a credit card and believe the product was misrepresented, you may contact your card issuer to dispute the charge. There are strict time limits (often 60-120 days from the transaction).
- Legal Action: For significant individual financial loss or medical injury, consult a consumer protection attorney. They can advise if your case merits a private lawsuit. Do not wait, as statutes of limitations apply.
Joining or Monitoring Class Action Litigation
As there is no current, certified class action, there is no “join” process. However, you can stay informed.
- Monitor Updates: Check reliable legal news sources or the websites of law firms that specialize in consumer class actions.
- Understand Certification: If a class action is filed in the future, a court must “certify” it, meaning it finds common legal questions outweigh individual differences. Only then would a process for class members to join or opt-out be established.
Red Flags: When to Consult a Lawyer
Consider seeking a legal consultation if you have experienced:
- Serious health injuries requiring medical treatment that you believe are linked to the product.
- Substantial financial loss as a distributor, with evidence that earnings were based on recruitment rather than legitimate retail sales.
- Specific evidence of intentional fraud or misrepresentation.
Expert Analysis: Legal and Medical Perspectives
Legal Expert Opinion: Regulatory Attorney Insights
From a regulatory law perspective, the FDA’s 2020 warning letter is significant. An attorney specializing in FDA law would note that while the violations were largely “technical” (labeling errors, reporting lapses), they are not trivial. Failure to report serious adverse events is a major red flag for the agency, suggesting potential gaps in a company’s safety vigilance. It often triggers more intense future scrutiny. The outcome of the 2017 pyramid scheme case—being forced into confidential arbitration—is a common and effective defense tactic for MLM companies. It limits public discovery, avoids a precedent-setting court ruling, and often reduces the leverage of plaintiffs.
Medical Expert Opinion: Supplement Safety Assessment
A physician or pharmacologist would emphasize that “isotonic delivery” is a marketing term, not a recognized medical or pharmacological category. The efficiency of nutrient absorption depends on many factors, including the specific nutrient, its formulation, and an individual’s physiology. They would stress that all supplements carry potential risks for interactions with medications, allergic reactions, or unintended side effects. The medical consensus is that consumers should prioritize a balanced diet and discuss any supplement use with a healthcare provider, who can assess individual need and risk.
Isotonix vs. Competitors: Safer Alternatives?
How Isotonix Compares to Standard Supplements
| Feature | Isotonix (MLM Brand) | Typical Retail/Online Supplement Brands |
|---|---|---|
| Price | Often higher due to MLM commission structure | Generally lower, more direct competition |
| Availability | Only through independent distributors | Widely available online, in stores |
| Third-Party Verification | No mention of USP, NSF, or ConsumerLab certification | Many brands seek these certifications for quality assurance |
| Transparency | History of FDA labeling violations | Varies, but many brands openly publish CoAs (Certificates of Analysis) |
Top Alternatives to Isotonix Products
When looking for alternatives, consider brands that prioritize:
- Third-Party Certification: Look for seals from USP, NSF International, or ConsumerLab.com. These indicate the product has been tested for ingredient accuracy, purity, and the absence of contaminants.
- Transparent Labeling: Brands that provide detailed information about ingredient sourcing and make batch testing results available.
- Retail Availability: Purchasing from established retailers or directly from reputable brands often means better pricing and standard consumer return policies.
The Future of Isotonix and Market America
Market America has publicly denied wrongdoing and stated its commitment to compliance. Following the FDA letter, the company likely implemented corrective actions, such as revising labels and adverse event reporting procedures. The broader future hinges on ongoing regulatory trends. The FTC and FDA are increasingly scrutinizing both deceptive health claims in the supplement industry and recruitment-focused practices in the MLM sector. Market America’s ability to demonstrate that its revenue comes primarily from product sales to genuine consumers, not from recruiting, will be key to avoiding future pyramid scheme allegations. For 2026-2027, the outlook is one of continued monitoring by regulators and informed skepticism from consumers.
Frequently Asked Questions
Is there a current class action lawsuit against Isotonix in 2026?
No, there is no major, active class-action lawsuit against Isotonix or Market America proceeding publicly in court as of early 2026. Past litigation has been resolved through private arbitration.
What did the FDA warning letter to Market America say?
The 2020 FDA letter cited Market America for failing to report two serious adverse events involving hospitalization and for misbranding multiple Isotonix products due to incorrect serving sizes, improper labeling formats, and other violations.
Are Isotonix supplements safe to take?
While many people use them without reported issue, the FDA’s action raises concerns about the company’s adverse event monitoring. Safety can vary by individual. Consult your doctor before starting any new supplement, especially if you have health conditions or take medications.
Is Market America a pyramid scheme?
A 2017 class-action lawsuit alleged it was an illegal pyramid scheme. The case was sent to confidential arbitration, so there is no public legal verdict. The FTC defines pyramid schemes by an emphasis on recruitment over product sales, which is the core of the allegations against Market America.
Can I get a refund for Isotonix products?
You must start with Market America’s official return policy. If you believe you were defrauded by false claims, you may attempt a chargeback with your credit card company or consult a lawyer regarding your options for seeking a refund.
What are the side effects of Isotonix OPC-3?
There is no definitive list of side effects. Anecdotal reports mention digestive issues, headaches, or allergic reactions. The FDA documented a serious adverse event report involving a product kit that included OPC-3, where a user was hospitalized.
How do I report a problem with Isotonix supplements?
Report any suspected adverse reaction to the FDA via its MedWatch program. This is the most important action you can take to contribute to public safety monitoring.
Key Takeaways and Action Steps
- Clarify the “Lawsuit”: Understand that the “Isotonix lawsuit” primarily refers to a 2020 FDA warning letter and a 2017 pyramid scheme class action that went to private arbitration. There is no active consumer class action in 2026.
- Recognize the FDA Findings: The FDA formally cited Market America for serious violations, including not reporting hospitalizations and having mislabeled products.
- Exercise Consumer Caution: Be highly skeptical of superlative health claims and “get-rich-quick” income promises associated with MLM supplements. Research independent, third-party certifications.
- Know Your Rights:
- Report adverse events to the FDA MedWatch.
- Keep all documentation (receipts, labels, communications).
- Consult professionals—a doctor before taking supplements, a lawyer for significant financial or health harm.
Disclaimer: This article is for informational purposes only and does not constitute legal or medical advice. For guidance on your specific situation, please consult with a qualified attorney or healthcare provider.
The story of Isotonix underscores a critical lesson for the modern wellness marketplace: vigilance is paramount. Regulatory actions and past litigation reveal a pattern of concerns over marketing claims, business practices, and regulatory compliance. As a consumer or potential distributor, your most powerful tools are skepticism, research, and a commitment to relying on verifiable information over promotional promises. Stay informed by monitoring official sources like the FDA and FTC, and prioritize transparency and evidence when making decisions about your health and finances.

